
DETAILED GUIDELINES ON CLASSIFICATION RULES FOR IN VITRO DIAGNOSTIC MEDICAL DEVICES (IVD)
Date Submitted: 30/01/2026 02:49 PM
Xem chi tiết
GUIDELINES FOR CLASSIFYING MEDICAL DEVICES (Circular 05/2022/TT-BYT): SPECIAL CLASSIFICATION RULES
Date Submitted: 28/01/2026 01:55 PM
Xem chi tiết
DETAILED GUIDANCE: RULES FOR CLASSIFICATION OF ACTIVE MEDICAL DEVICES UNDER CIRCULAR 05/2022/TT-BYT
Date Submitted: 26/01/2026 01:57 PM
Xem chi tiết
GUIDELINES FOR CLASSIFICATION OF INVASIVE MEDICAL DEVICES (According to Circular No. 05/2022/TT-BYT)
Date Submitted: 23/01/2026 02:49 PM
Xem chi tiết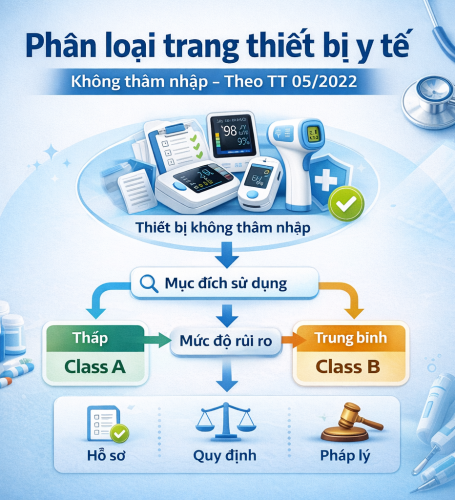
DETAILED GUIDELINES: CLASSIFICATION RULES FOR NON-INVASIVE MEDICAL DEVICES (In accordance with Circular No. 05/2022/TT-BYT)
Date Submitted: 21/01/2026 02:43 PM
Xem chi tiết
CLASSIFICATION OF MEDICAL DEVICES According to Decree No. 98/2021/ND-CP & Circular No. 05/2022/TT-BYT
Date Submitted: 19/01/2026 11:48 AM
Xem chi tiết
Declaration of Applied Standards – Mandatory Procedure for Class A & B Medical Devices
Date Submitted: 16/01/2026 02:02 PM
Xem chi tiết
GUIDELINES ON DECLARATION OF APPLICABLE STANDARDS FOR MEDICAL DEVICES
Date Submitted: 14/01/2026 10:53 AM
Xem chi tiết
DETAILED GUIDELINES ON IMPORT PROCEDURES FOR MEDICAL DEVICES
Date Submitted: 12/01/2026 04:19 PM
Xem chi tiết
LATEST IMPORT PROCEDURES FOR DIETARY SUPPLEMENTS & FULL-SERVICE SOLUTIONS FROM SONGWIN LOGISTICS
Date Submitted: 09/01/2026 11:58 AM
Xem chi tiết











